Vocal Cord Paralysis, Unilateral
Unilateral vocal cord paralysis is the neurogenic inability of one vocal cord to move. It is associated with weak voice of a degree that can vary between individuals.
See also: vocal cord paralysis, bilateral.
What Causes Vocal Cord Paralysis?
If the recurrent laryngeal nerve gets damaged anywhere along the way from brainstem to larynx, then some or all of the vocal cord muscles might be weakened (paretic) or completely immobilized (paralyzed), thereby affecting the person’s voice or breathing.
Can the Nerve Recover on Its Own?
If the damaged nerve is still intact, then it may recover on its own, either completely or at least enough to meet the person’s vocal needs. This recovery can take up to a year because neural tissue heals much more slowly than skin or even bone.
Sometimes, though, the nerve does not recover, or not enough to meet the person’s vocal needs. There is no need to wait for a potential recovery if a tumor has invaded the nerve or if the nerve is known to be severed.
Symptoms
One of the most common scenarios is that after investigation the cause is unknown—we call these cases idiopathic. In these cases, a virus is suspected to have played a role.
Two other causes of paresis or paralysis are tumors and trauma. Tumors in these cases could be in the thyroid, lung, esophagus, etc. Trauma is often from surgery that is performed along the path of the vagus nerve, such as for thyroid, spine, lung, or heart problems.
Other symptoms may include:
- Weak, air-wasting dysphonia – The voice may be breathy, weak, double-pitched, or manifest luffing. The person may only be able to say a few words on a breath or be unable to project the voice in noisy places.
- Inability to be heard in noisy locations.
- A tendency of the voice to be somewhat stronger in the morning but to “fade” with use.
- A tendency to cough when drinking thin liquids.
Testing That May Be Done
Laryngeal videostroboscopy
When performed at close range, this examination helps to determine which muscles are affected.
CT scan
This test helps to rule out mass lesions along the course of the nerve. A CT scan is not necessary if the cause is already known (e.g., thyroid surgery).
EMG
This is optional/controversial, and not needed in clinics where a close-range laryngeal videostroboscopy is performed.
Treatment Options
Do nothing
Just accept the symptoms while waiting, especially if recovery is thought to be possible (such as after thyroid surgery with an intact nerve, or after a presumed viral injury).
Vocal exercise with a speech pathologist
Usually helpful if the patient is taciturn. Some patients are mistakenly advised (by well-intentioned family or friends) to rest their voices. Voice rest is in fact counterproductive, except when the general fatigue caused by use of an air-wasting voice makes brief voice rest appropriate.
Voice gel – an injected implant that is slowly absorbed and therefore temporary
This is not for definitive voice restoration, but can offer a boost while awaiting possible recovery. The aim is for modest or better benefit.
Voice gel implants are often done in-office with topical anesthesia and, occasionally, mild sedation. The gel material—hyaluronic acid—infiltrates the tissue to plump and firm so that the other vocal cord has something to push against.
Duration of benefit is typically four to twelve weeks as the gel slowly absorbs. The hope is that nerve recovery occurs during this time, but injection can be repeated if its benefit fades before it is appropriate to place a permanent implant.Voice gel + hydroxyapatite — A semi-permanent injected implant
This option is used mostly if the patient does not want a medialization laryngoplasty (option #5 below). If placement of the filler is not ideal, a surgical adjustment might be necessary.
Medialization laryngoplasty with a silastic implant
This is performed in an outpatient operating room under local anesthesia with deep sedation (“twilight”). Surgery requires a horizontal one-inch incision on the front of the neck, and involves placement of a wedge or “shim” to permanently medialize and firm up the paralyzed vocal cord.
There are small risks of bruising and infection. Approximately one out of ten patients needs a second trip to the operating room to adjust the implant.
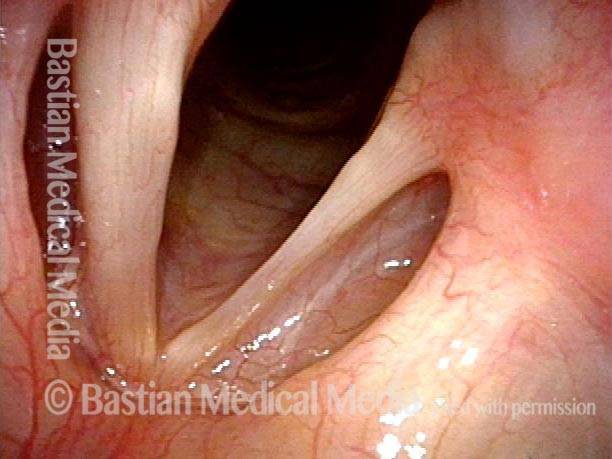
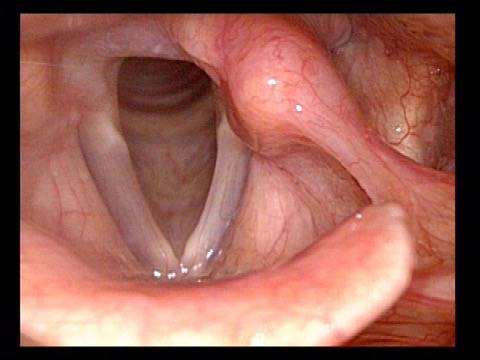
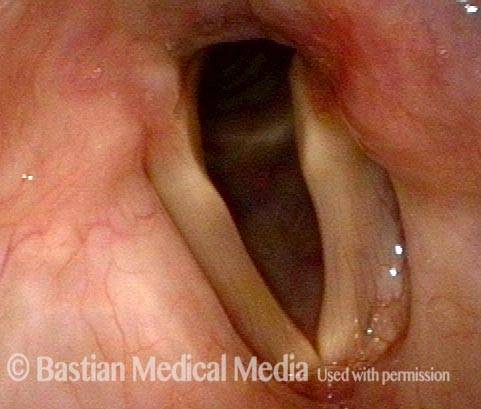
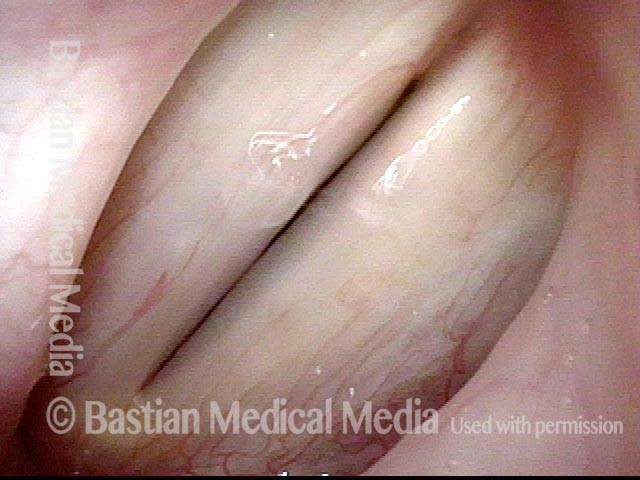
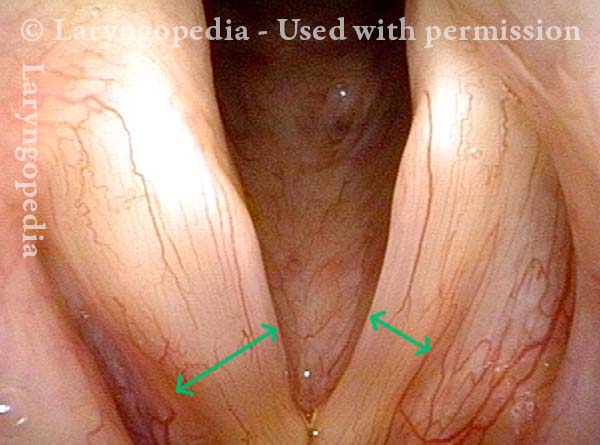
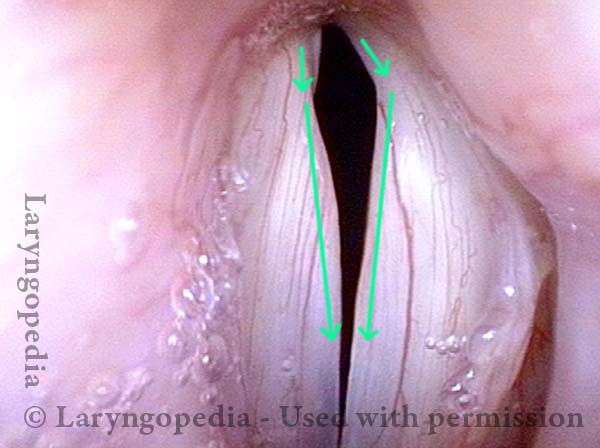
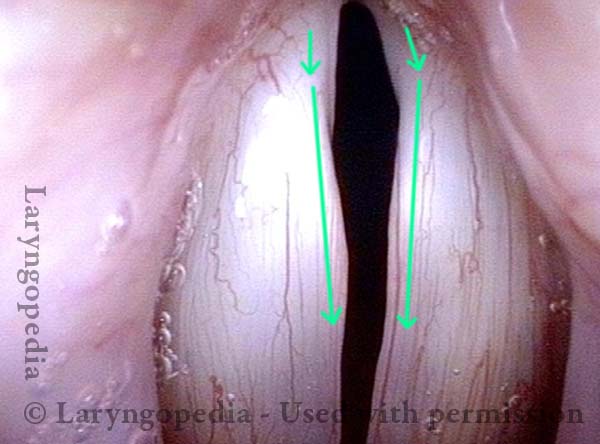
Vocal Cord Paralysis, Before and After Medialization
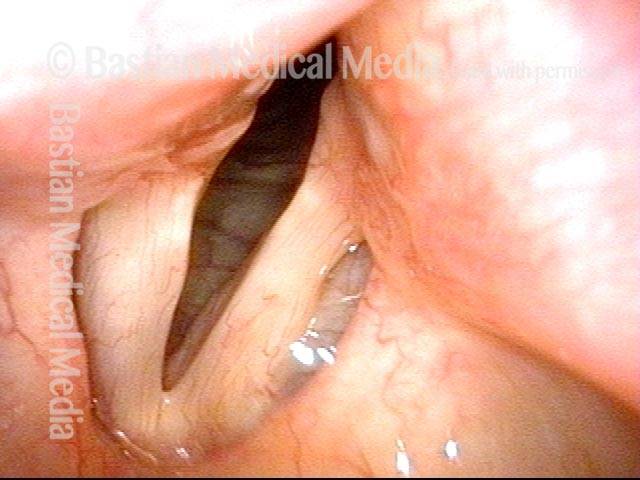
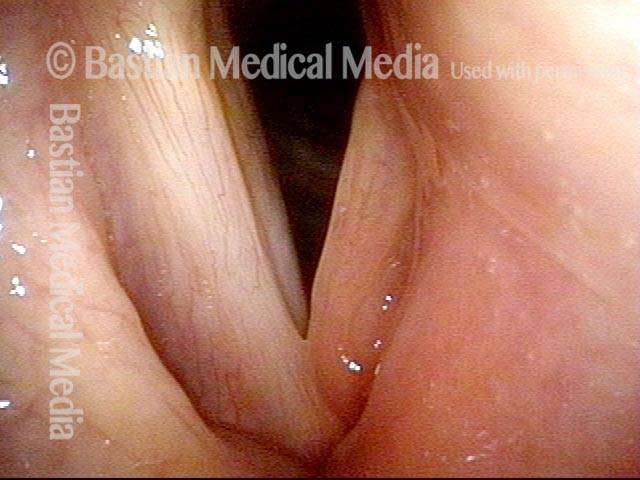
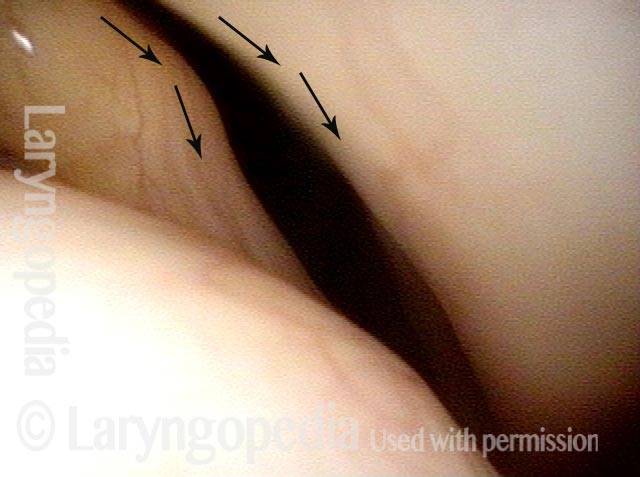
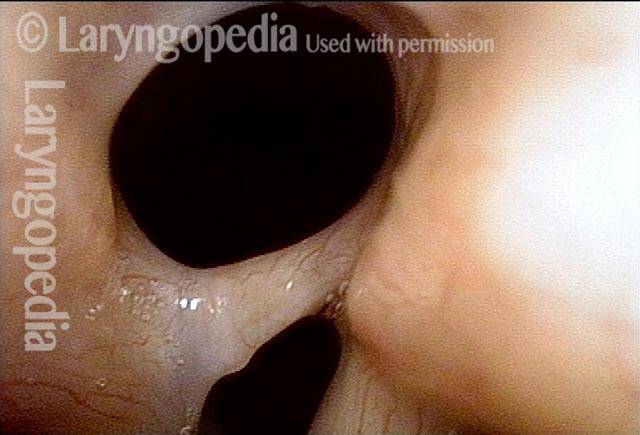
Vocal cord paralysis: before medialization (1 of 12)
Vocal cord paralysis: before medialization (1 of 12)
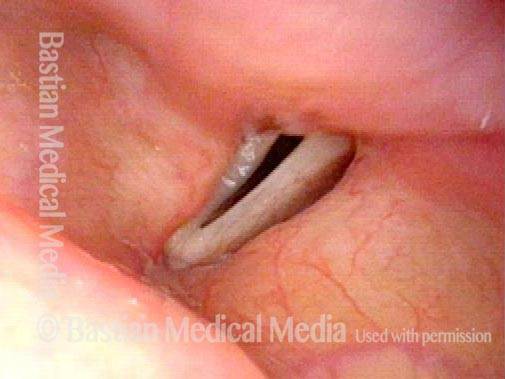
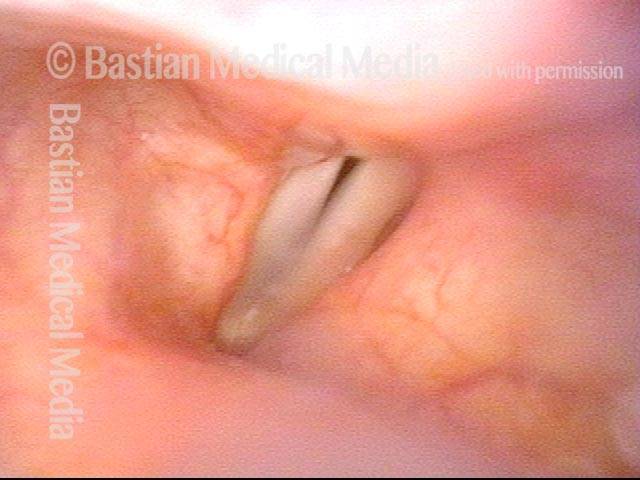
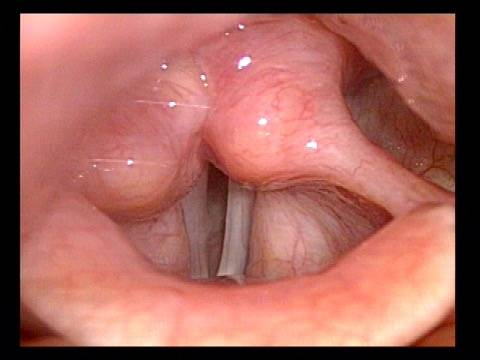
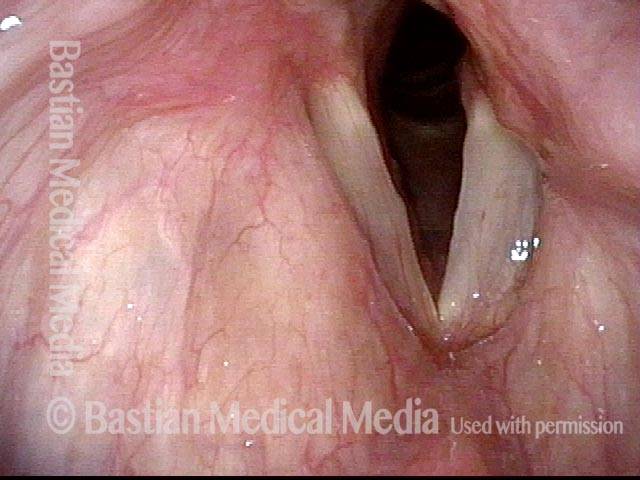
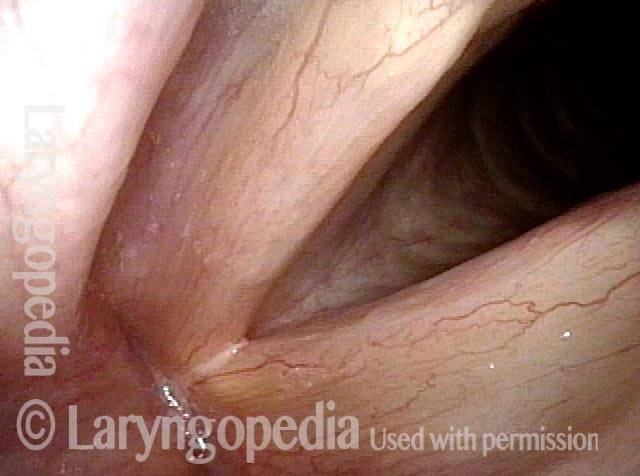
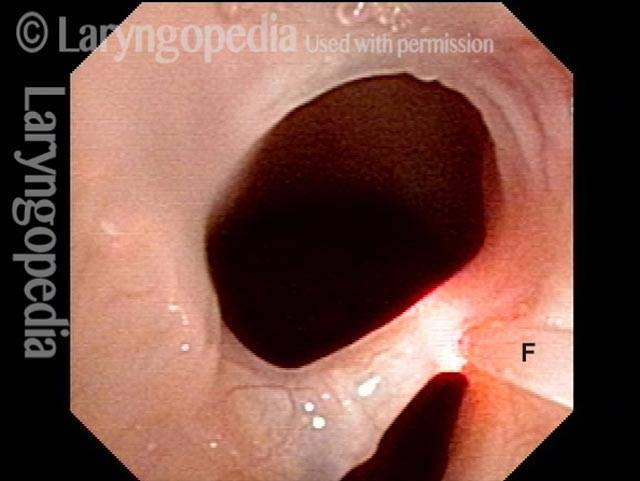
Vocal cord paralysis: before medialization (2 of 12)
Vocal cord paralysis: before medialization (2 of 12)
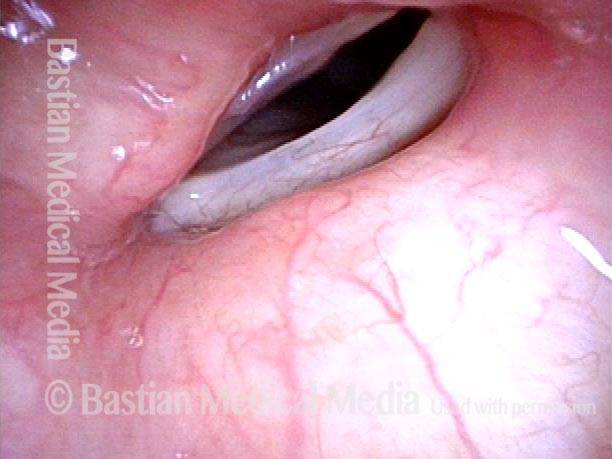
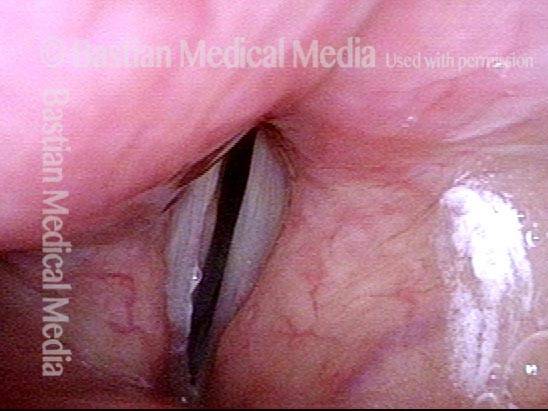
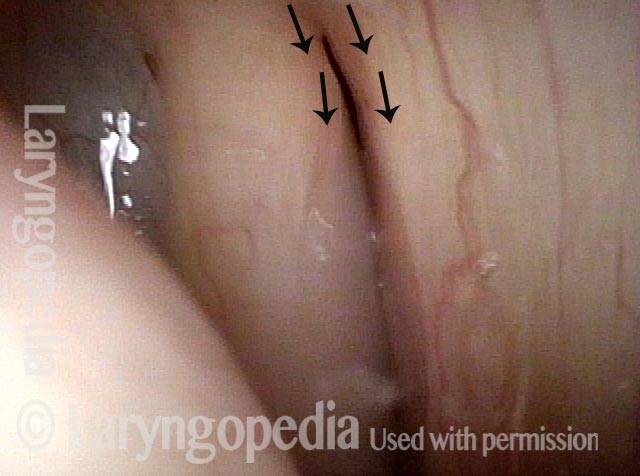
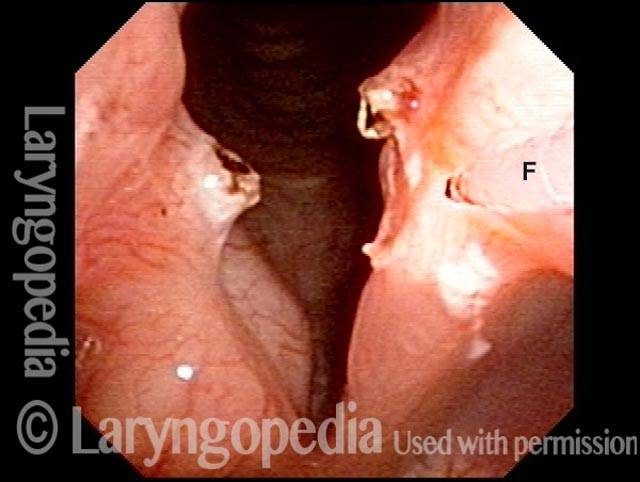
Vocal cord paralysis: before medialization (3 of 12)
Vocal cord paralysis: before medialization (3 of 12)
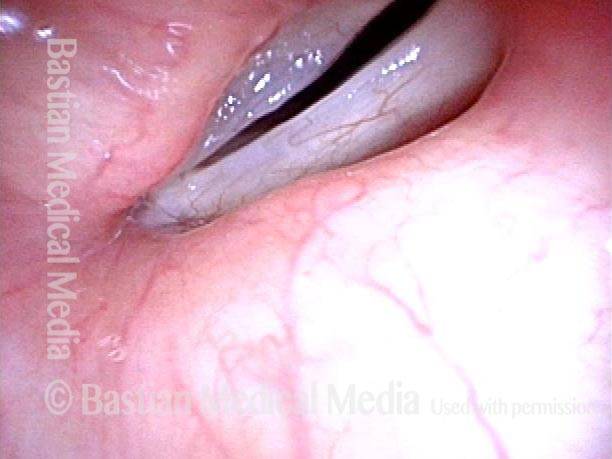
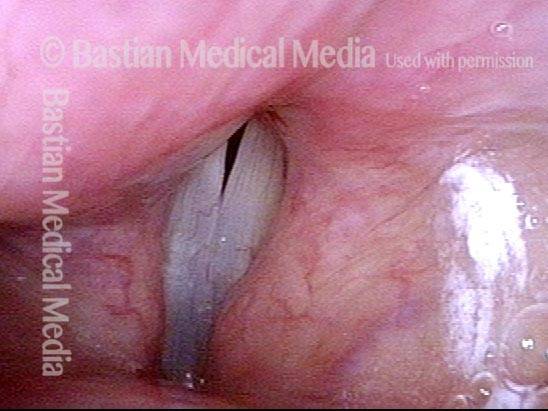
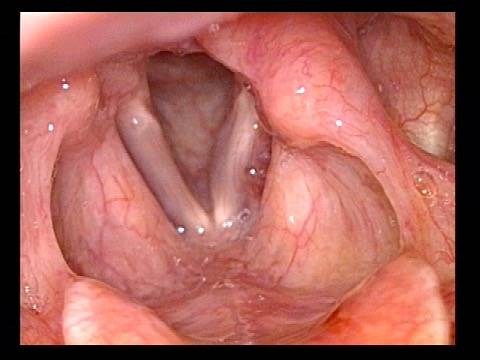
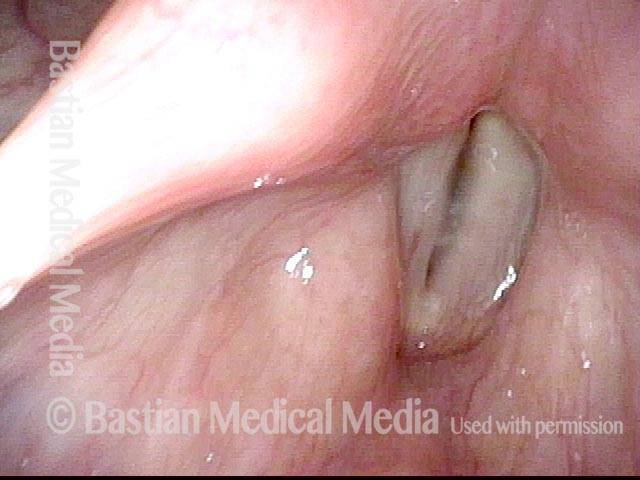
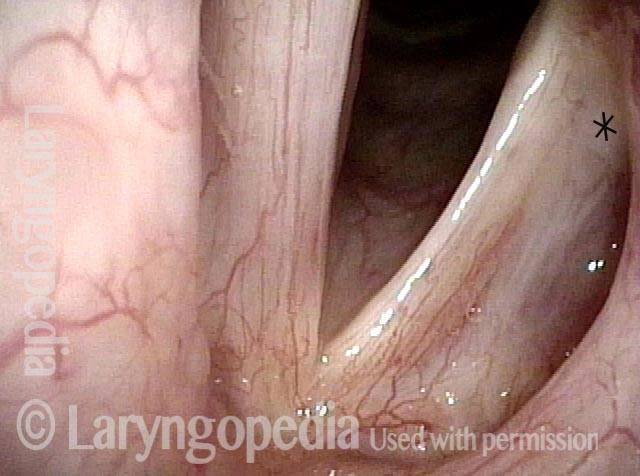
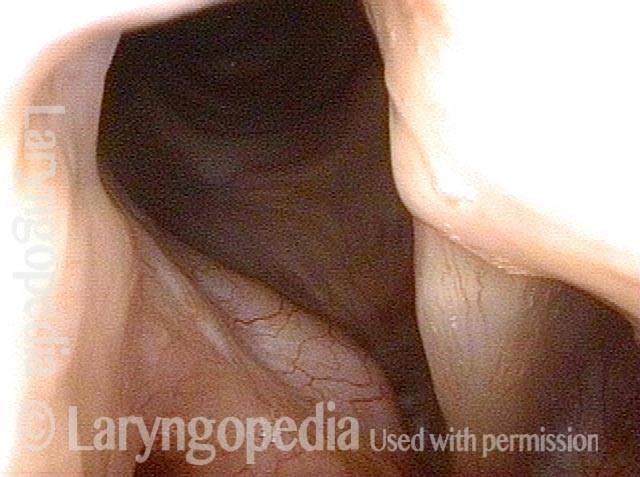
Vocal cord paralysis: before medialization (4 of 12)
Vocal cord paralysis: before medialization (4 of 12)
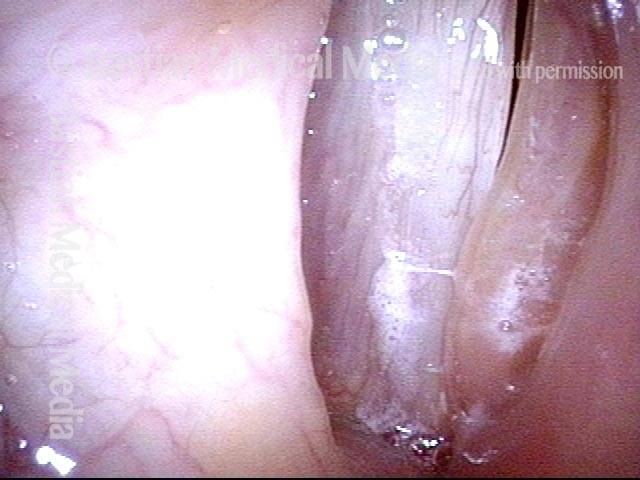
Vocal cord paralysis: 1 week after medialization (5 of 12)
Vocal cord paralysis: 1 week after medialization (5 of 12)
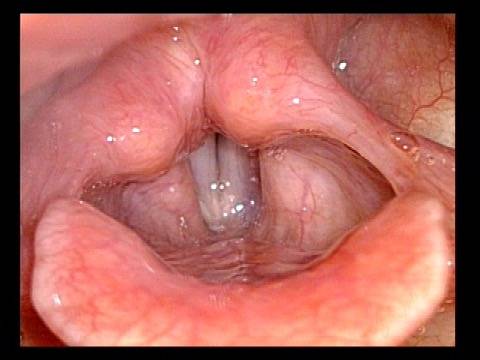
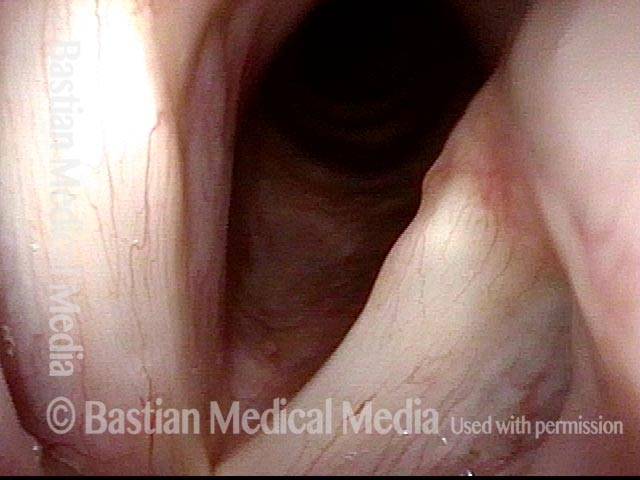
Vocal cord paralysis: 1 week after medialization (6 of 12)
Vocal cord paralysis: 1 week after medialization (6 of 12)
Vocal cord paralysis: 1 week after medialization (7 of 12)
Vocal cord paralysis: 1 week after medialization (7 of 12)
Vocal cord paralysis: 1 week after medialization (8 of 12)
Vocal cord paralysis: 1 week after medialization (8 of 12)
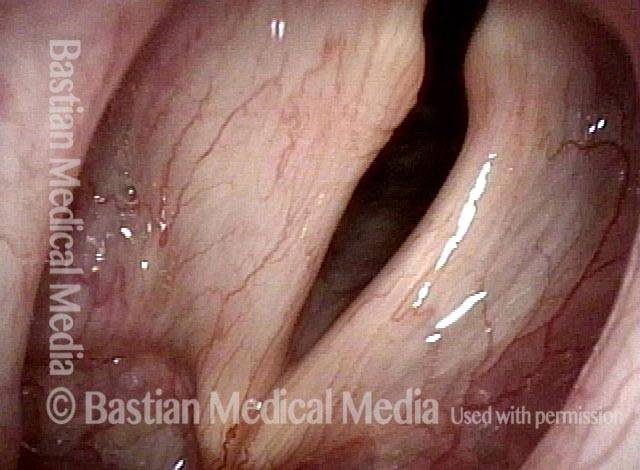
Vocal cord paralysis: 5 months after medialization (9 of 12)
Vocal cord paralysis: 5 months after medialization (9 of 12)
Vocal cord paralysis: 5 months after medialization (10 of 12)
Vocal cord paralysis: 5 months after medialization (10 of 12)
Vocal cord paralysis: 5 months after medialization (11 of 12)
Vocal cord paralysis: 5 months after medialization (11 of 12)
Vocal cord paralysis: 5 months after medialization (12 of 12
Vocal cord paralysis: 5 months after medialization (12 of 12
What to expect after Medialization
- Do I need to rest my voice? No. Just use it “prudently” for a few days.
- How long till my voice recovers? The voice will be hoarse for a few days to weeks before it “settles.” In light of this, most patients take a few days to a week off of work.
- How much improvement can I expect? A good result is going from 20% to 70-80%, of normal voice, but this is not guaranteed.
- What if I’m unhappy with my result? Sometimes we wait for further evolution; sometimes we try a few sessions of speech therapy; sometimes we do a revision surgical procedure. This is all according to the patient’s needs and other details.
Injection Laryngoplasty with Temporary Gel
Laryngoplasty (1 of 4)
Laryngoplasty (1 of 4)
Reason for air-wasting (2 of 4)
Reason for air-wasting (2 of 4)
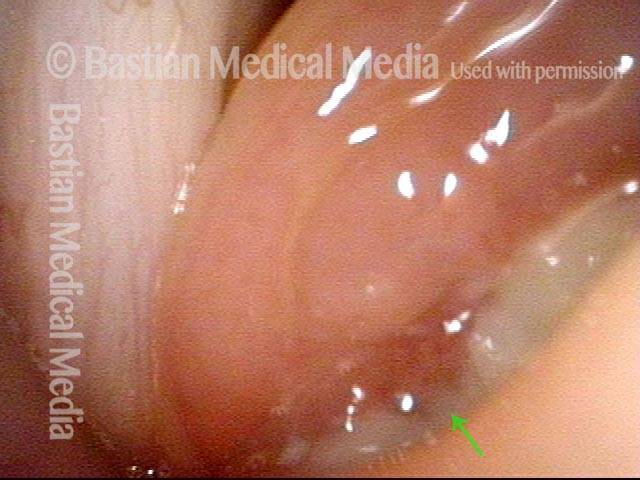
Voice gel injected into vocal cord (3 of 4)
Voice gel injected into vocal cord (3 of 4)
Vocal cords close completely (4 of 4)
Vocal cords close completely (4 of 4)
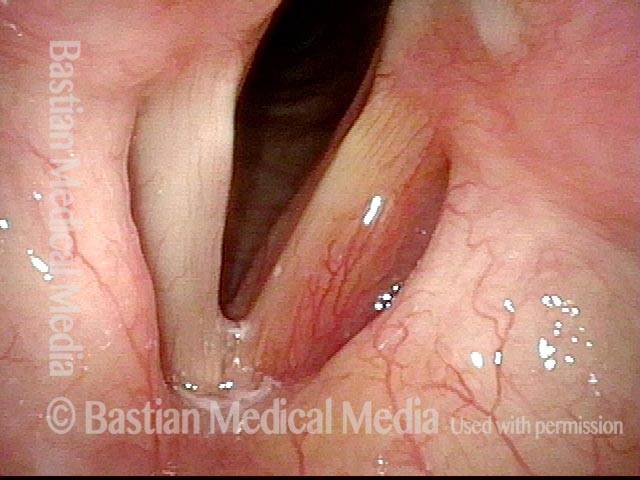
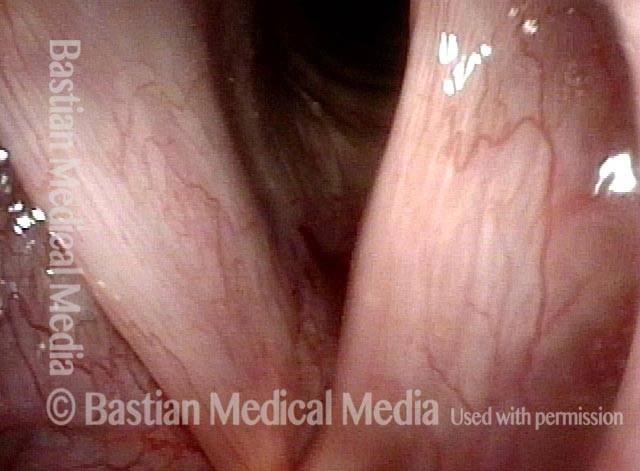
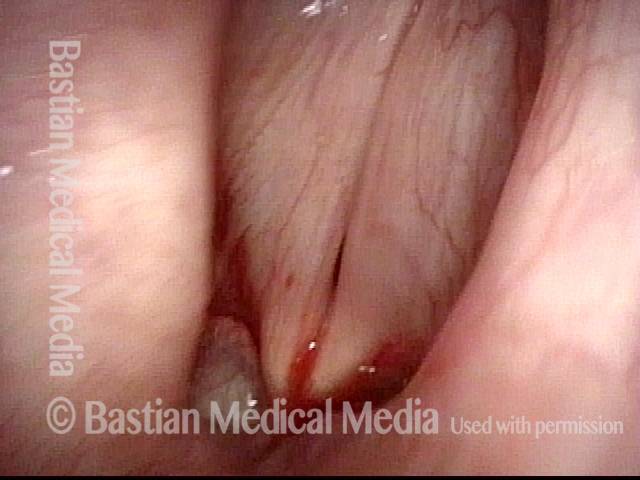
Vocal Cord Paralysis
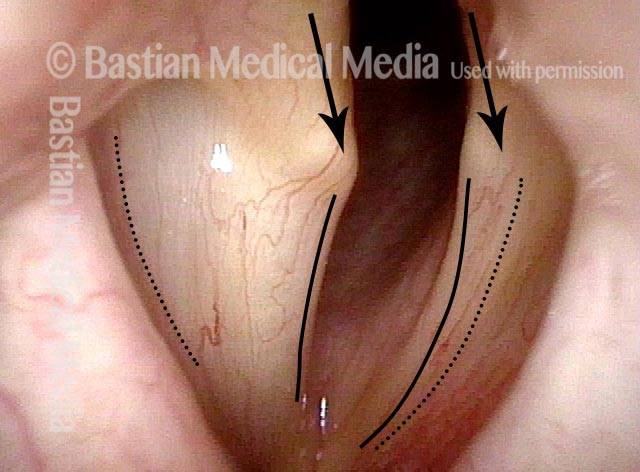
Vocal cord paralysis (1 of 2)
Vocal cord paralysis (1 of 2)
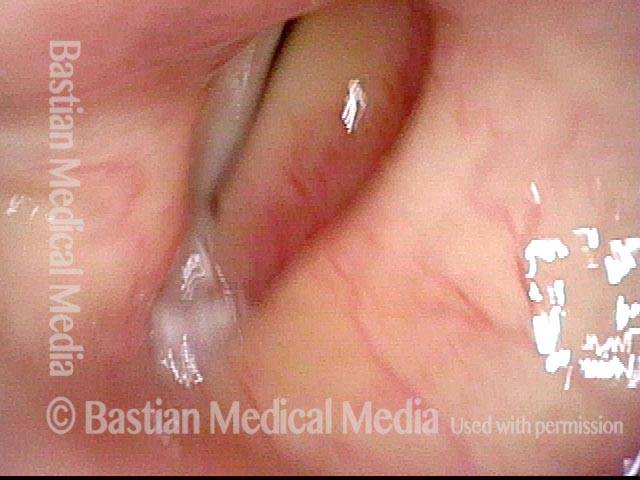
Vocal cord paralysis (2 of 2)
Vocal cord paralysis (2 of 2)
Example 2
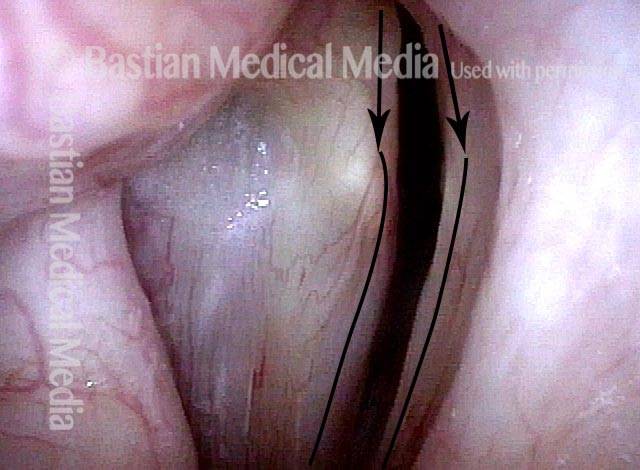
Vocal cord paralysis (1 of 5)
Vocal cord paralysis (1 of 5)
Phonation (2 of 5)
Phonation (2 of 5)
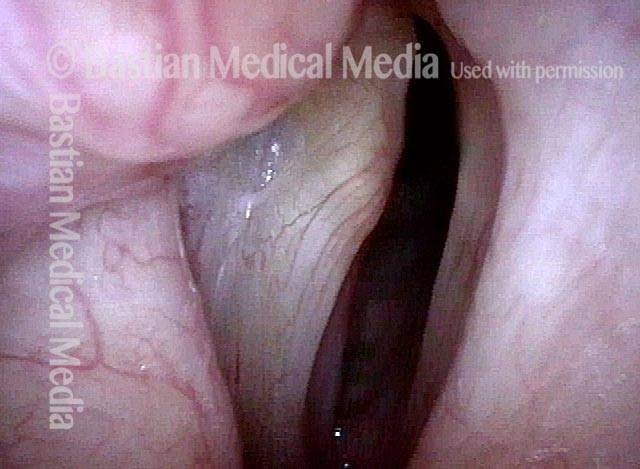
Closer view (3 of 5)
Closer view (3 of 5)
Evidence of LCA weakness (4 of 5)
Evidence of LCA weakness (4 of 5)
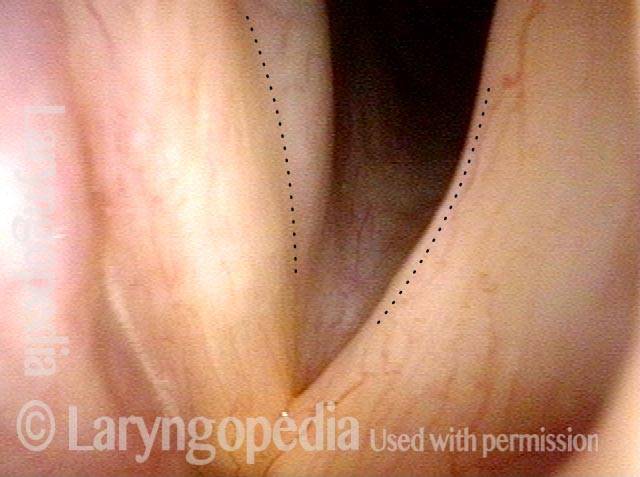
Phonation at very high pitch (5 of 5)
Phonation at very high pitch (5 of 5)
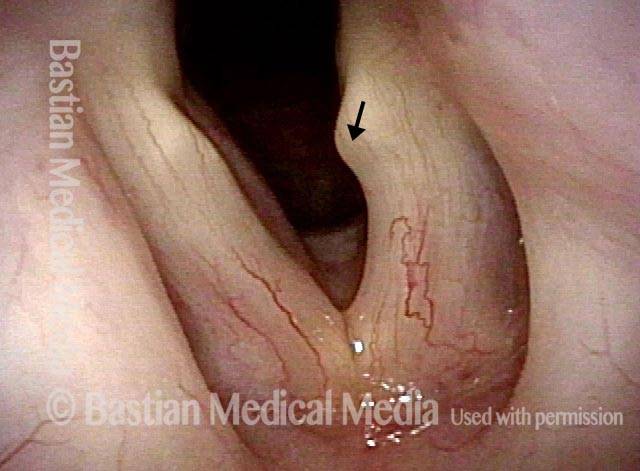
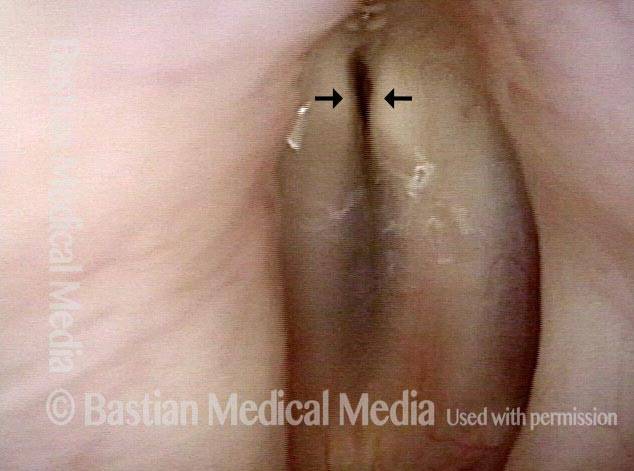
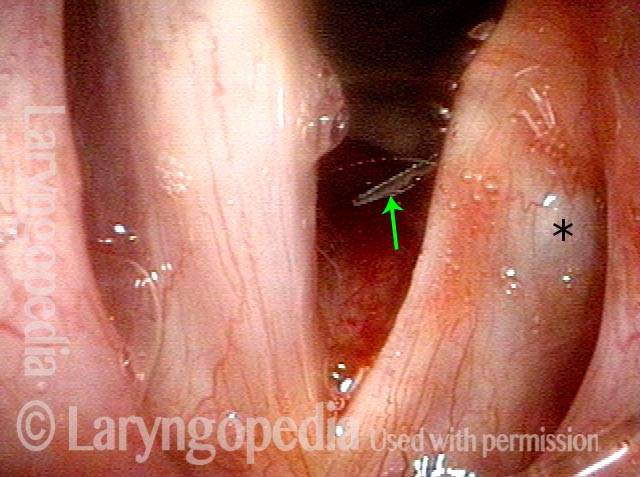
Paralysis, Anterior Branch Recurrent Nerve
Spaghetti vs linguini folds (1 of 3)
Spaghetti vs linguini folds (1 of 3)
Denervation of TA + LCA (2 of 3)
Denervation of TA + LCA (2 of 3)
Anterior stretch (3 of 3)
Anterior stretch (3 of 3)
Extrusion of Vocal Cord Implant
Extrusion of vocal cord implant (1 of 3)
Extrusion of vocal cord implant (1 of 3)
Extrusion of vocal cord implant (2 of 3)
Extrusion of vocal cord implant (2 of 3)
Extrusion of vocal cord implant (3 of 3)
Extrusion of vocal cord implant (3 of 3)
Voice Gel for Immediate Help
Paralysis of left vocal cord (1 of 4)
Paralysis of left vocal cord (1 of 4)
Vocal gel injection (2 of 4)
Vocal gel injection (2 of 4)
Cord closer to midline (3 of 4)
Cord closer to midline (3 of 4)
Complete adduction of cords (4 of 4)
Complete adduction of cords (4 of 4)
TA + PCA-only Paresis
Breathing position (1 of 5)
Breathing position (1 of 5)
Phonatory position (2 of 5)
Phonatory position (2 of 5)
Voice gel injection (3 of 5)
Voice gel injection (3 of 5)
Plumped vocal cord (4 of 5)
Plumped vocal cord (4 of 5)
After voice gel injection (5 of 5)
After voice gel injection (5 of 5)
55 Years of Paralysis with Every Classic Finding
Breathing position, bowed left cord (1 of 4)
Breathing position, bowed left cord (1 of 4)
Approaching voicing position (2 of 4)
Approaching voicing position (2 of 4)
Vocal cord closure, large gap (3 of 4)
Vocal cord closure, large gap (3 of 4)
Open phase of vibration, flaccidity of right cord (4 of 4)
Open phase of vibration, flaccidity of right cord (4 of 4)
Medialization Laryngoplasty Typically Doesn’t Fix the LCA “Finding.”
Left vocal cord paralysis (1 of 4)
Left vocal cord paralysis (1 of 4)
Bowing during phonation (2 of 4)
Bowing during phonation (2 of 4)
After medialization (3 of 4)
After medialization (3 of 4)
Lateral turning remains unchanged (4 of 4)
Lateral turning remains unchanged (4 of 4)
Voice Gel Injection for Vocal Cord Paralysis
Left vocal cord paralysis (1 of 4)
Left vocal cord paralysis (1 of 4)
Voice-making position (2 of 4)
Voice-making position (2 of 4)
Voice gel injection (3 of 4)
Voice gel injection (3 of 4)
After voice gel injection (4 of 4)
After voice gel injection (4 of 4)
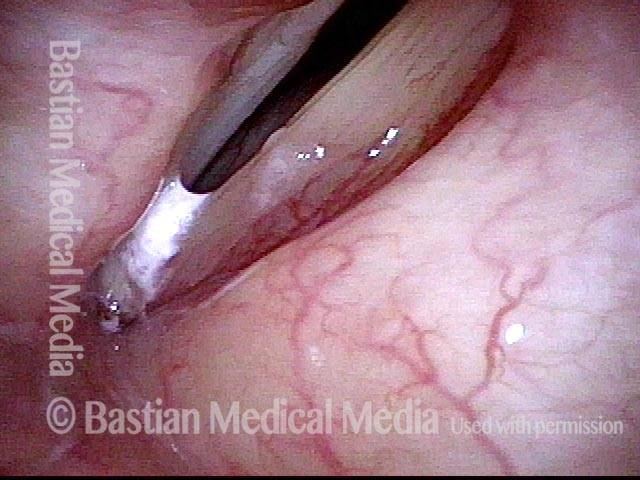
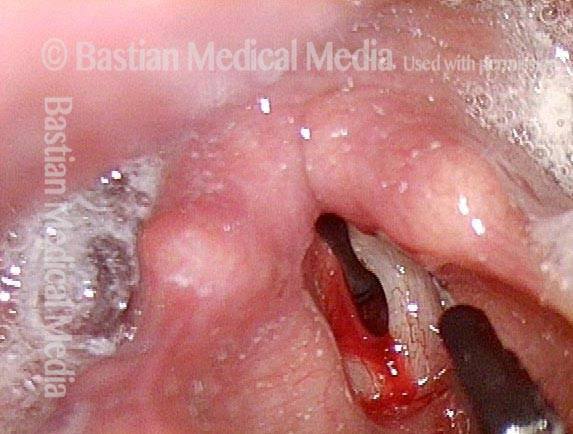
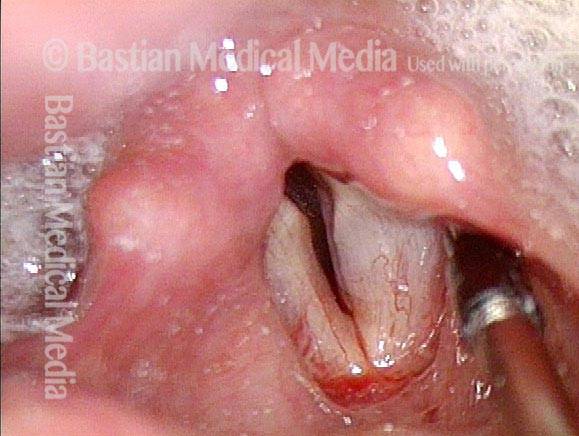
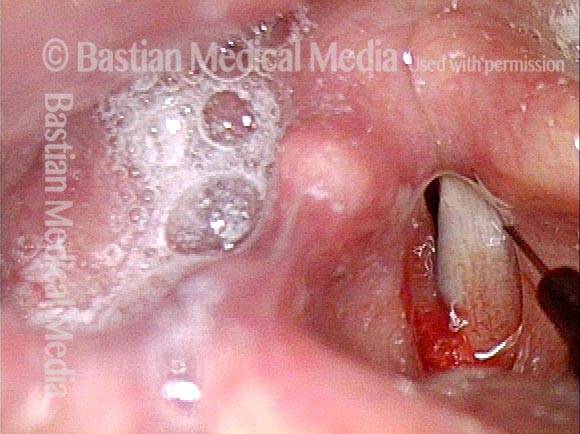
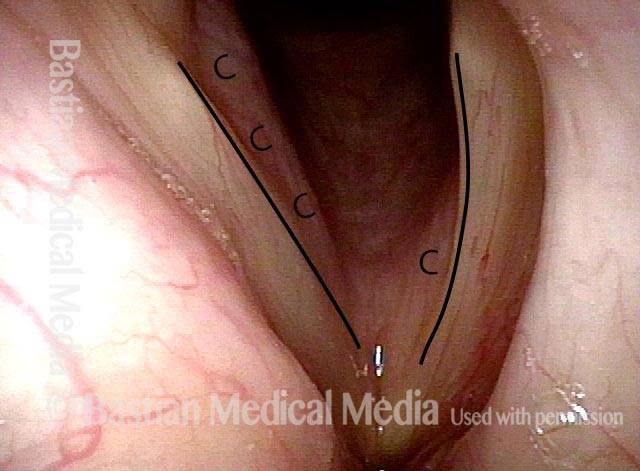
Posterior Commissure Synechiae
Tethered vocal cords (1 of 5)
Tethered vocal cords (1 of 5)
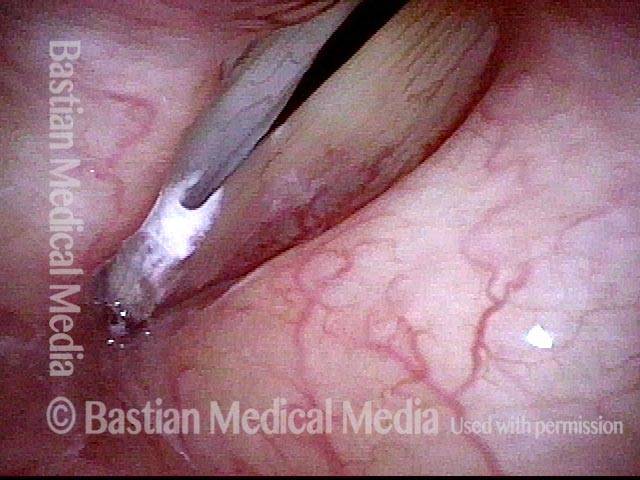
Before laser removal (3 of 5)
Before laser removal (3 of 5)
Immediately after laser (4 of 5)
Immediately after laser (4 of 5)
One month post-op (5 of 5)
One month post-op (5 of 5)
Do I Need Surgery for Vocal Cord Paralysis?
There is unfortunately not a lot one can do to speed healing. In the photo essay Vocal Cord Paresis Accentuated by Disuse, the patient had sufficient spontaneous recovery to give her an “adequate” voice. The main thing is NOT to rest the voice as this woman did, as all that does is to atrophy any muscle that remains functional. And one can try “voice building” to see if it adds any strength.
If those things don’t work and it is 9 months or more since the onset of the paralysis, the only options are to accept the “new” voice or to proceed to medialization.
Keep in mind that one type of medialization is a simple injection, and the other is surgery through a one-inch incision.