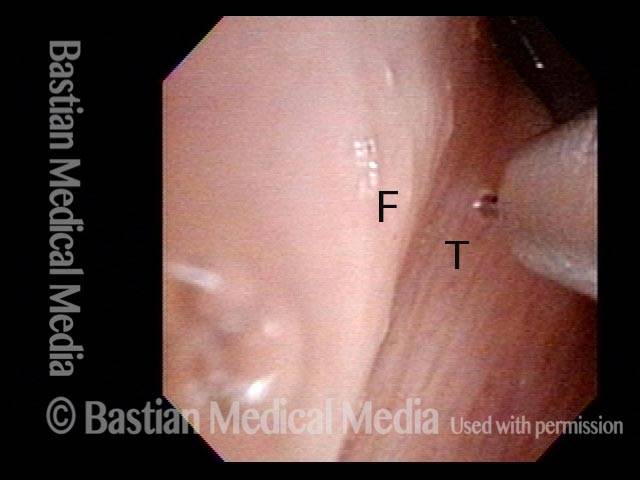
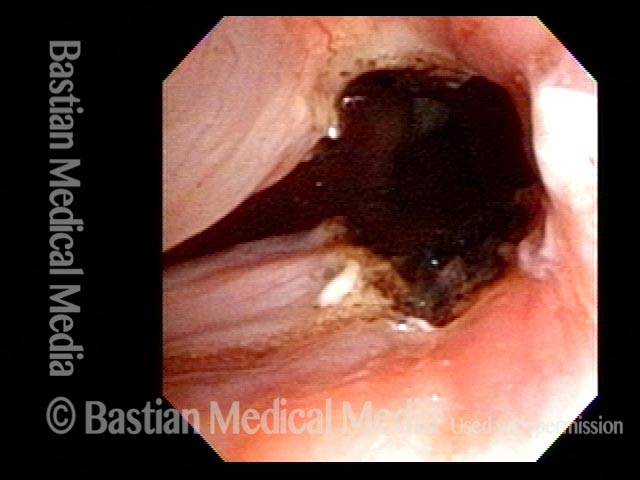
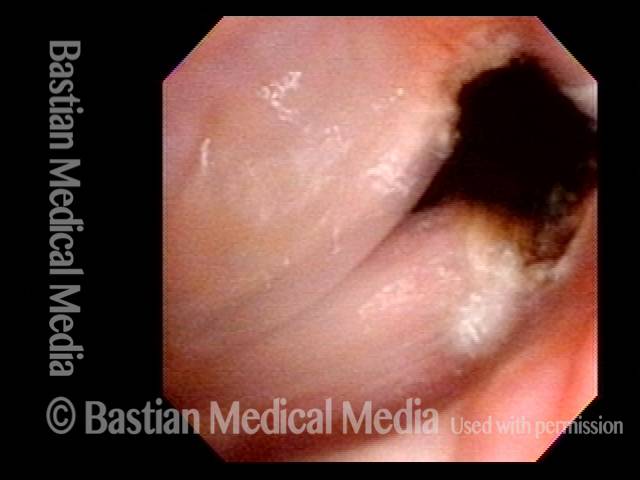
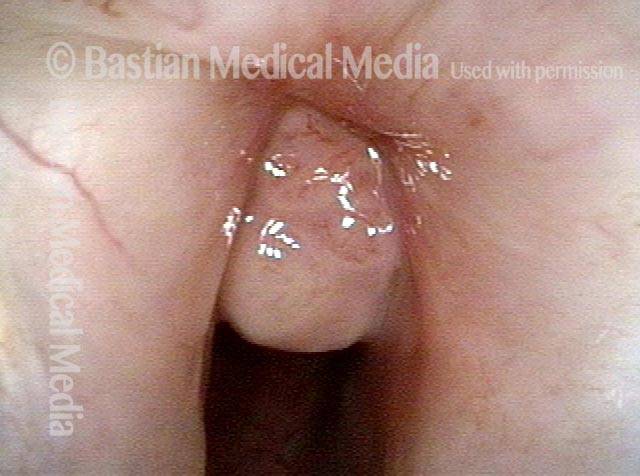
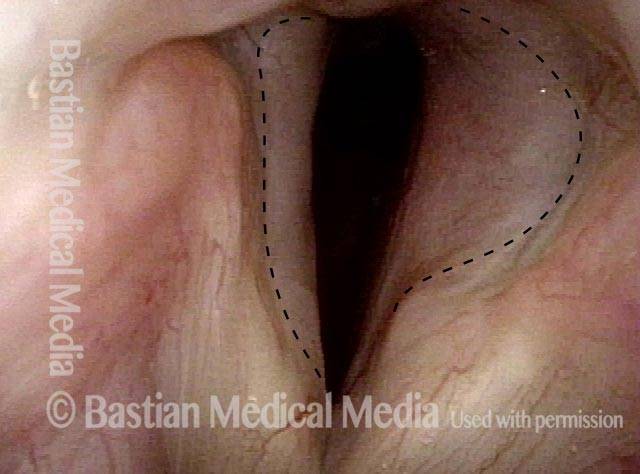
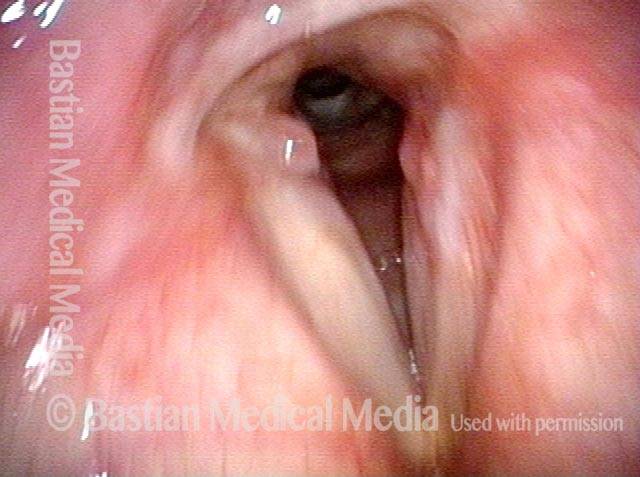
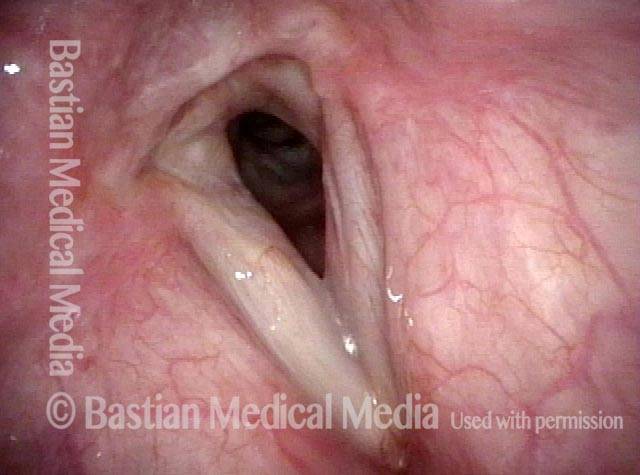
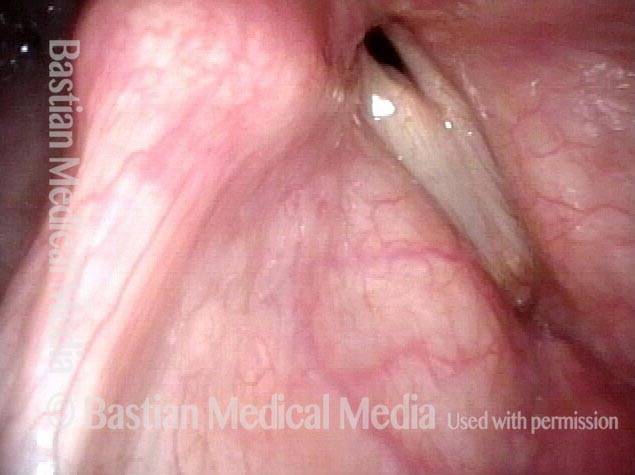
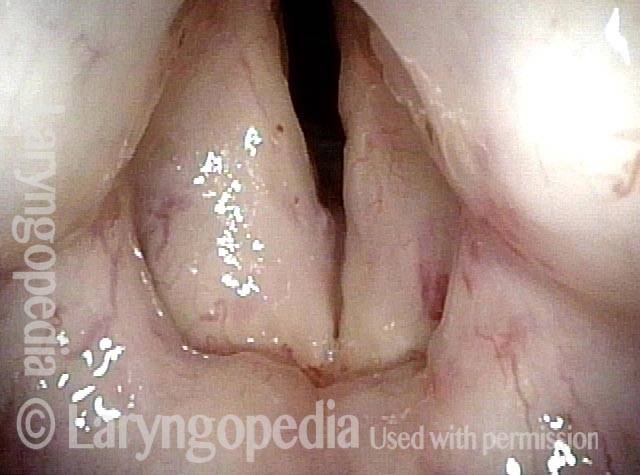
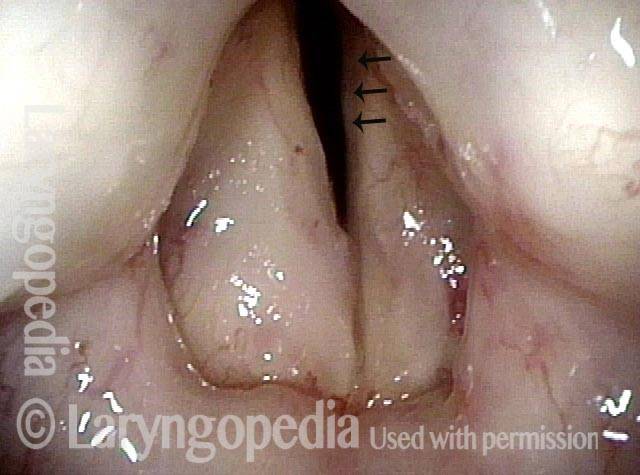
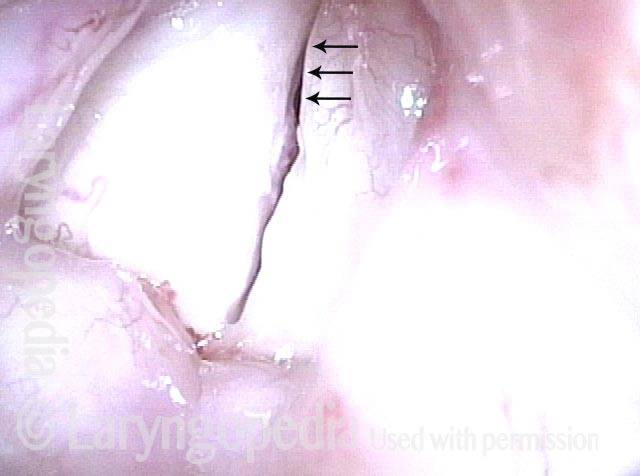
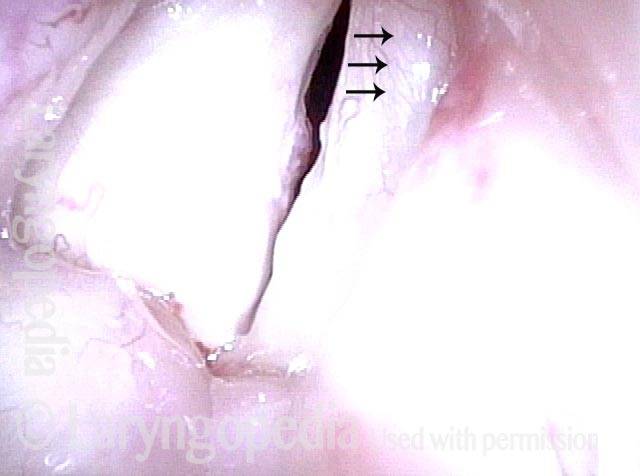
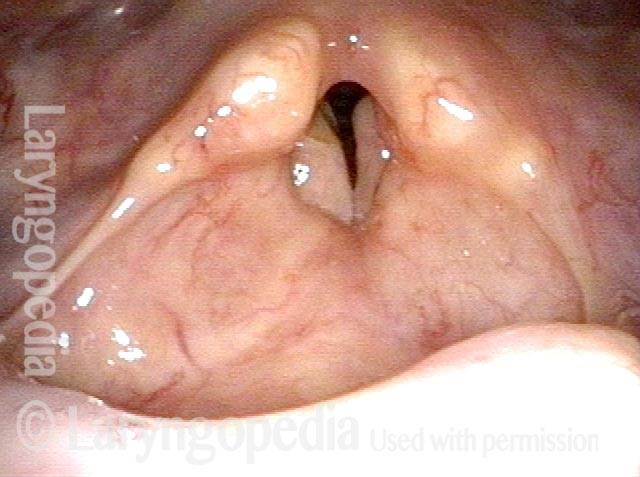
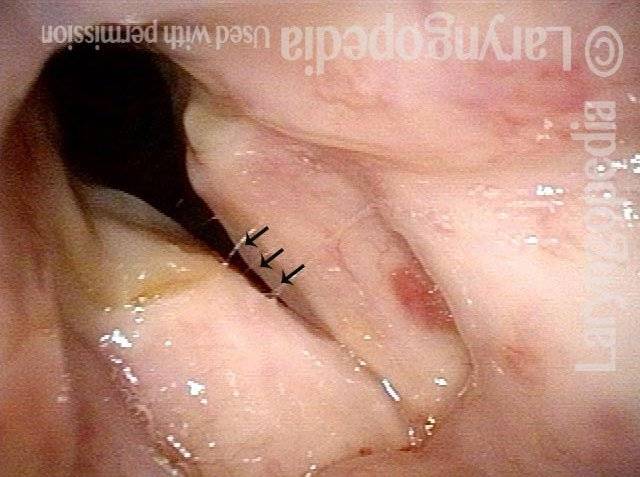
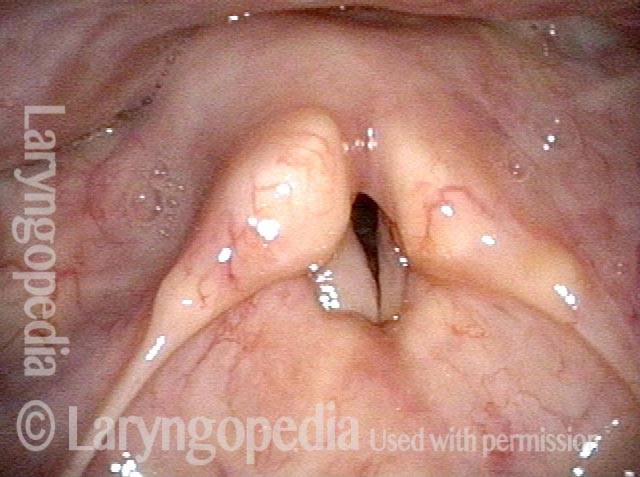
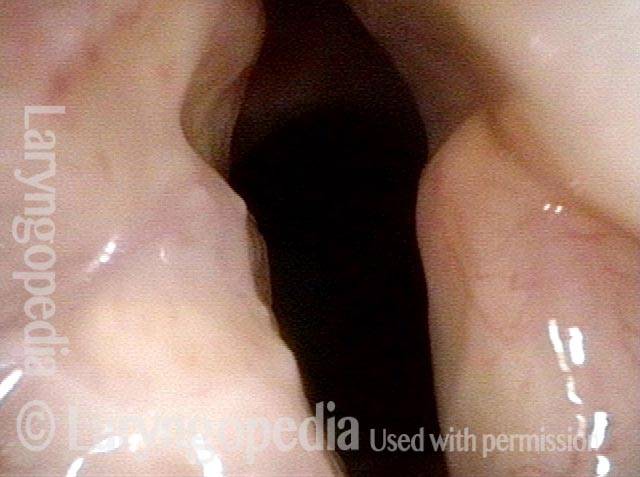
Posterior commissuroplasty is an endoscopic procedure performed for individuals who have difficulty breathing, due to either bilateral vocal cord paralysis or bilateral vocal cord fixation. These individuals’ vocal cords are immobile or fixed in a mostly closed position, which inhibits breathing and often causes noisy inspiration.
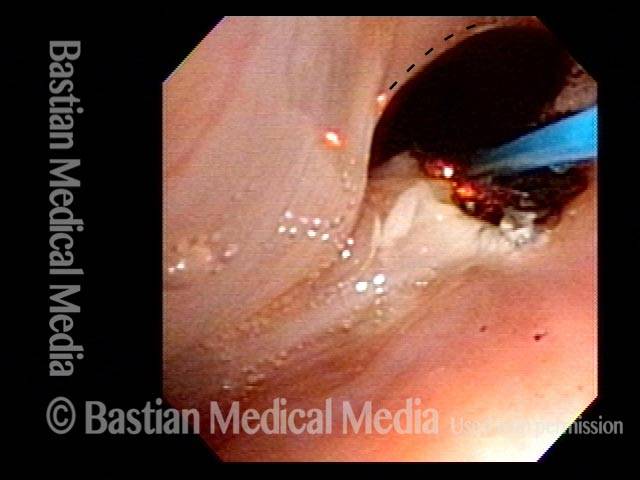
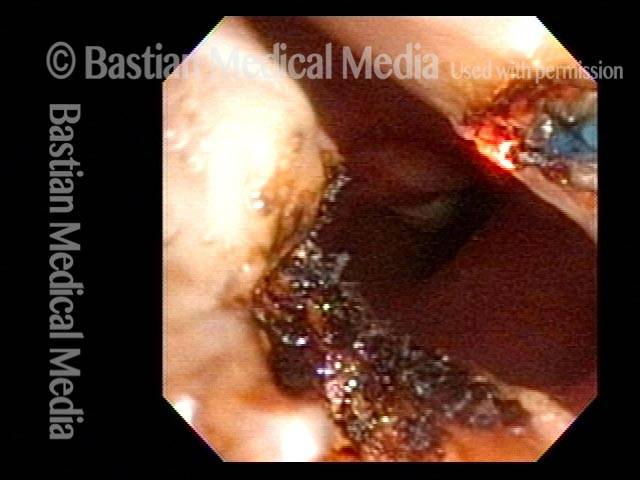
In a posterior commissuroplasty, the clinician uses a carbon dioxide laser to take small divots from the posterior ends (membranous glottis) of both vocal cords. These divots create more space between the cords so that, during breathing, air can pass through more easily. This procedure can avoid the need for a tracheotomy. It also preserves the voice’s functionality better than a transverse cordotomy typically does.